Due to their energy differences, soft X-ray and hard X-ray absorption spectroscopy excel at surface detection and bulk analysis, respectively. They complement each other in fields like materials science and biology, working together to reveal the structural and chemical states of matter. Light is a form of energy generated by electron movement; different wavelengths appear as different colors, even though most wavelengths are invisible to the human eye.

Figure 1: Light/Waves
01. What is X-Ray Absorption Spectroscopy?
When exploring the microscopic world, X-ray Absorption Spectroscopy (XAS) acts as an incredibly powerful analytical tool.
Imagine it as a pair of "eyes" capable of peering into the hidden mysteries of matter. By measuring how much X-rays are absorbed as they pass through a sample, XAS reveals the material's electronic structure, the local arrangement of its atoms, and the chemical state of its elements.
The magic behind this technique happens when energy-tunable X-ray photons interact with matter. They excite core-level electrons within the atoms, causing them to transition and create an absorption "signal" at specific energy points. Together, these signals form a spectrum—much like a unique "fingerprint" for the element being tested, packed with rich physical and chemical information.
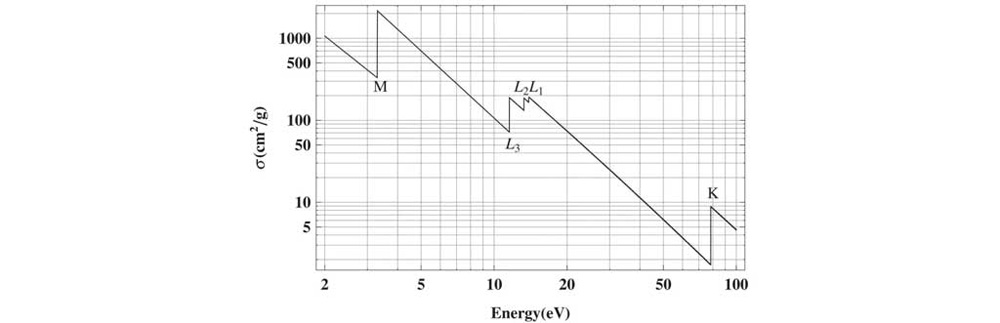
Figure 2: Schematic of an Absorption Spectrum
XAS technology generally consists of two main parts: X-ray Absorption Near Edge Structure (XANES) and Extended X-ray Absorption Fine Structure (EXAFS).
The former focuses on the fine structure right around the absorption edge, making it extremely sensitive to the chemical valence state and the symmetry of the coordination environment. The latter analyzes the oscillating signals extending beyond the absorption edge, which can accurately deduce structural information such as the types of coordinating atoms surrounding the central atom, the coordination number, and bond lengths.
Of course, these "X-ray eyes" operate in two distinct modes: soft X-rays and hard X-rays. X-rays are categorized into these two realms based on their energy levels (or wavelengths). In this article, We will dive deep into the differences between soft and hard X-ray absorption spectroscopy, exploring how they team up to build a complete analytical framework,Relying on the motion control system equipped with granite base and high-precision guide rail and lead screw.
02. Basic Principles and Core Differences: Energy Dictates Perspective
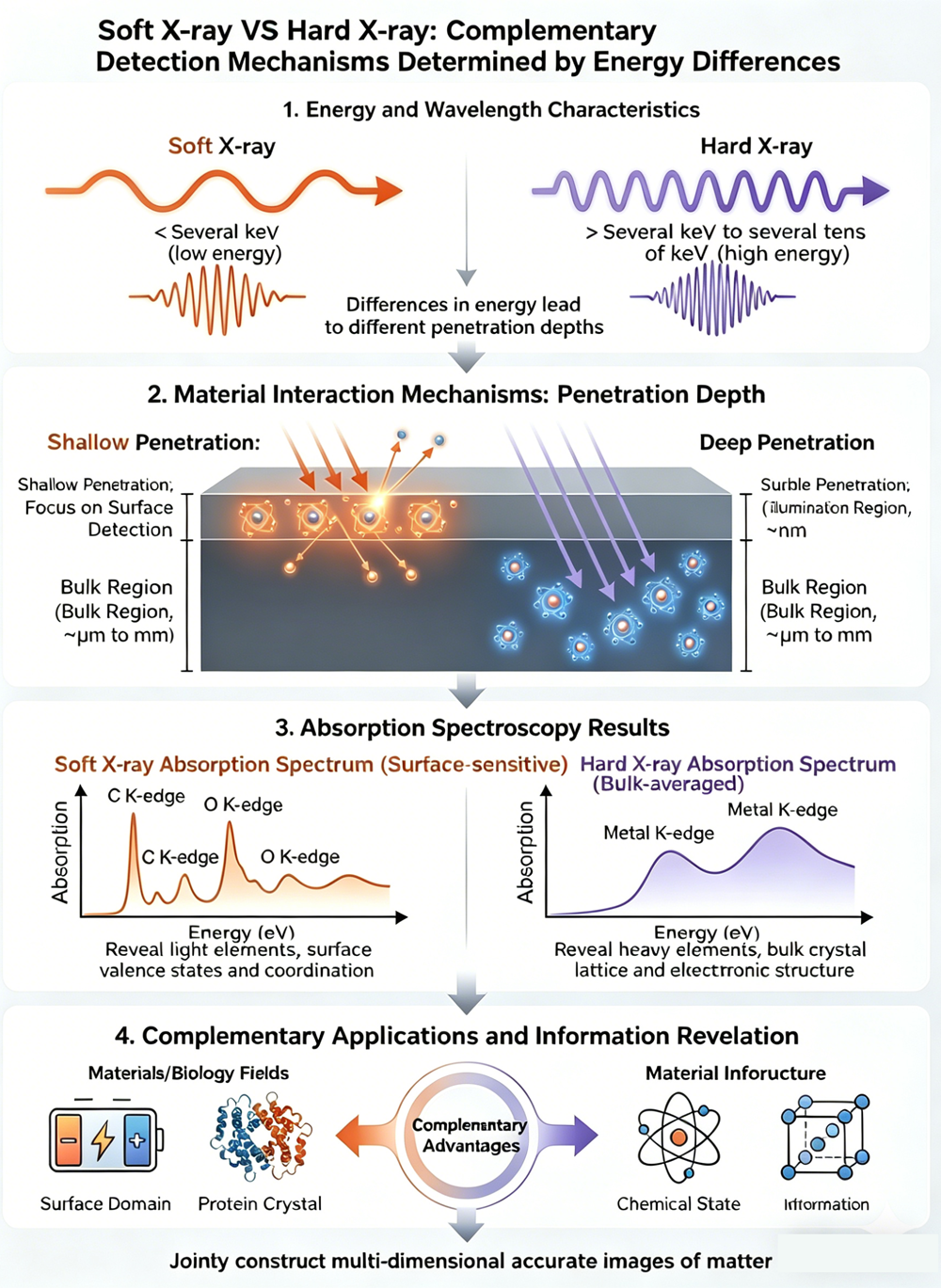
The most fundamental difference between soft and hard X-ray spectroscopy stems from their energy ranges. This single factor directly dictates their physical properties and detection capabilities.
The Divide of Energy and Wavelength
Soft X-rays: Generally refer to X-rays with lower energy and longer wavelengths. Their energy ranges roughly from 200 electron volts (eV) to 2,000 eV, with corresponding wavelengths of about 10 to 100 Angstroms (Å).
Hard X-rays: These boast higher energy and shorter wavelengths. Their energy typically exceeds 5 kilo-electron volts (keV), with wavelengths shorter than 1 Å.
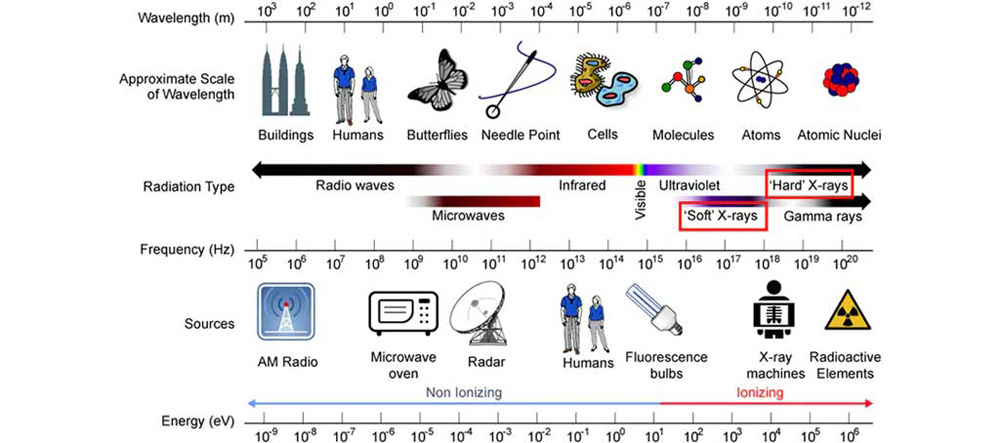
Figure 3: Schematic of X-ray Wavelengths
As energy increases, the X-ray absorption coefficient smoothly drops, meaning the penetrating power of the X-rays significantly increases. This very law of physics creates the core difference in how they probe matter: penetration depth.
Penetration Depth: Surface vs. Bulk
Soft X-rays: The "Microscope" of the Surface World
Because they have lower energy, soft X-rays are easily absorbed when interacting with matter, severely limiting their penetrating power. In the lab, soft XAS is essentially a surface-probing technique.
For instance, in Total Electron Yield (TEY) mode, its probing depth is merely a few nanometers—perfect for capturing the chemical information of a sample's outermost layer. Even in the slightly more penetrating Fluorescence Yield (FY) mode, the depth is only between 150 nanometers and 1 micrometer. This extreme surface sensitivity makes soft X-rays the ideal tool for studying surfaces, thin films, and the interfacial chemical and electronic structures of materials.
Hard X-rays: The "Probe" for the Deep Interior
In stark contrast, hard X-rays pack high energy and low absorption coefficients, giving them robust penetrating power. They can effortlessly slice through samples tens of micrometers or even millimeters thick to gather structural and chemical information that represents a statistical average of the sample's interior.
Therefore, hard XAS is highly suited for studying bulk materials, powder catalysts, or materials sealed within specific reaction environments (like batteries or reaction cells), providing an overall view of the bulk material's chemical state and atomic coordination.
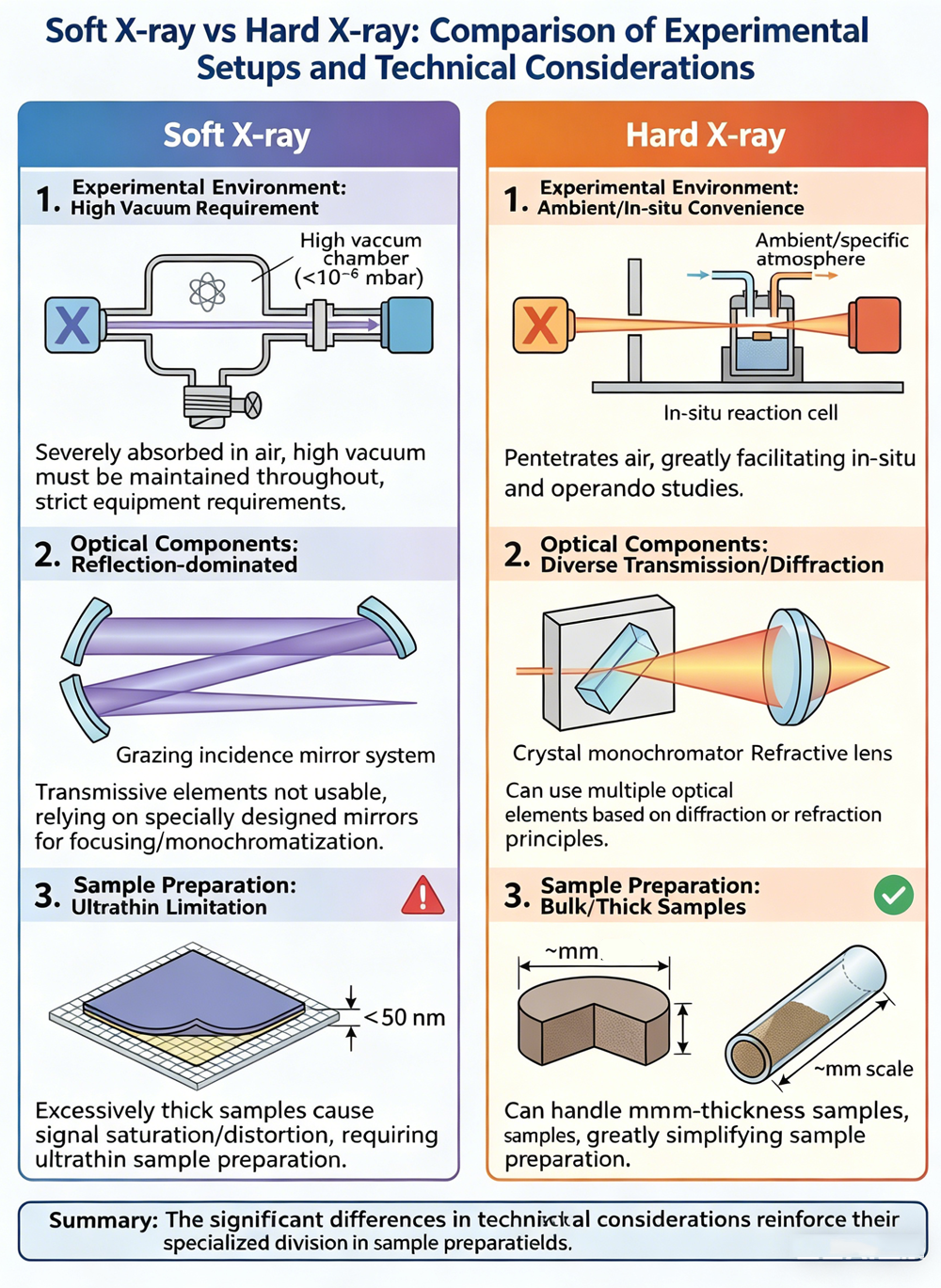
03. Experimental Setup and Technical Considerations: Different "Rules of the Game"
Because soft and hard X-rays differ so vastly in their physical nature, setting up their experiments, preparing samples, and managing measurement environments require playing by completely different "rules of the game."
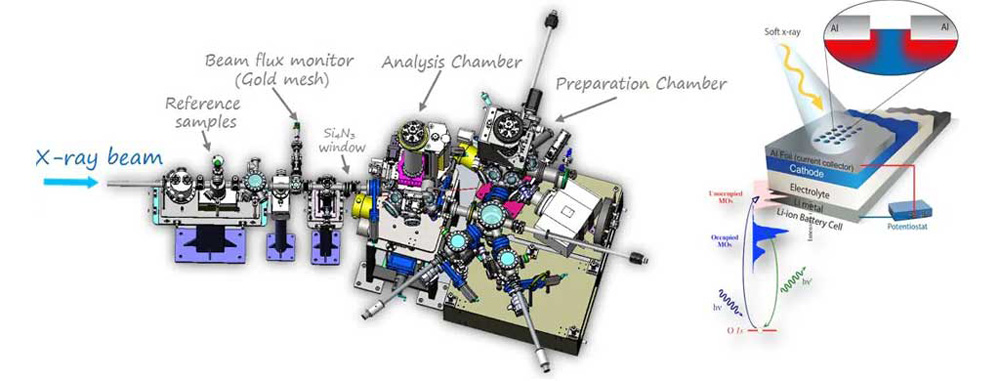
Figure 4: SSRF BL02B02 Beamline - Soft X-ray Experimental Platform

Figure 5: SSRF BL17B X-ray Absorption Fine Structure Spectroscopy - Hard X-ray Experimental Platform
Vacuum Requirements: Soft X-rays are heavily absorbed by air, rendering the atmosphere practically opaque to them. Consequently, the entire optical path—from the light source to the sample and finally to the detector—must be maintained in a high-vacuum environment, placing immense demands on the equipment. Hard X-rays, however, can easily pass through air. Their experiments can be conducted under atmospheric conditions or specific gas environments, offering incredible convenience for in-situ and operando studies.
Optical Differences: The transmissive optical components we commonly use for visible light, such as lenses and prisms, are completely useless in the soft X-ray spectrum. Focusing and monochromatizing soft X-rays rely entirely on specially designed reflective mirror systems. Hard X-rays, on the other hand, can utilize a variety of optical components based on diffraction (like crystal monochromators) or refraction.
Sample Preparation Constraints: When measuring with soft X-rays, there are strict limits on sample thickness. If a sample is too thick (e.g., over 50 nanometers), it can cause the absorption signal to saturate or distort, ruining the spectral data. Conversely, hard X-ray experiments can handle samples up to millimeters thick, drastically simplifying the sample preparation process.
These technical distinctions further reinforce the specialized roles that soft and hard XAS play in their respective application fields.
04. Application Areas: Complementary Scientific Perspectives
Though soft and hard X-ray absorption spectroscopy share the same underlying principles, their different targets and penetration depths allow them to play complementary roles, pushing the boundaries of scientific discovery.
Target Elements and Absorption Edges
The energy range of Soft X-rays makes them the weapon of choice for studying the K-edge absorption spectra of low-Z elements—such as Carbon (C), Nitrogen (N), and Oxygen (O), which are the core building blocks of life sciences and organic materials.
Hard X-rays, meanwhile, are primarily used to probe the K-absorption edge of transition metals and heavier elements (exciting electrons from the 1s orbital to the 4p orbital). The K-edge EXAFS signal provides precise structural information about the coordination environment surrounding the central atom (like bond lengths and coordination numbers). It is the gold standard for pinpointing the active sites of catalysts, the central structures of metalloproteins, and lattice distortions in materials.
Unique Applications in Life Sciences
Hard X-rays are widely used in biology to study metalloenzymes and metalloproteins. By analyzing the K-edge absorption spectra of metal cofactors like iron, copper, and zinc in proteins, scientists can track their valence changes and the dynamic restructuring of their coordination environments during biological catalytic cycles, thereby unraveling how enzymes actually work.
Soft X-rays open a highly unique window in biological imaging. In a specific energy range known as the "water window" (roughly 280 eV to 543 eV, nestled between the K-edges of carbon and oxygen), water absorbs soft X-rays relatively weakly, while carbon-rich organic matter (like proteins and lipids) absorbs them strongly. This natural contrast allows soft X-ray microscopes to capture high-resolution images of intact cells in a watery, near-natural state, revealing their intricate internal structures.
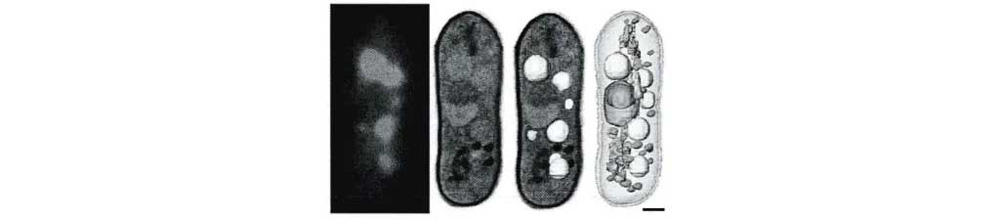
Figure 6: Cryo-fluorescence of yeast cell vacuoles and the corresponding soft X-ray tomographic reconstruction and segmented rendering
05. Emerging Trends and Future Outlook
With the rapid advancement of cutting-edge light sources like modern synchrotrons and X-ray Free-Electron Lasers (XFEL), both soft and hard XAS technologies are stepping into a new era of possibilities.This advancement also drives the development of higher-precision motion control systems, which require more stable and reliable granite component bases to support their operation.
Time-Resolved Tracking of Dynamic Processes: By harnessing the ultra-fast, ultra-intense X-ray pulses provided by XFELs, scientists are developing time-resolved XAS technologies. Think of it as "shooting a movie" to track transient structural and electronic changes during chemical reactions, phase transitions, or the execution of biological macromolecule functions in real time.
Deepening In-Situ and Operando Studies: Researchers are designing increasingly sophisticated in-situ reaction cells. By combining the high-penetration power of hard X-rays with the surface sensitivity of soft X-rays, scientists can now study the dynamic evolution of materials under realistic operating conditions (such as high temperatures, high pressures, or complex electrochemical environments), successfully bridging the gap between fundamental research and practical applications.
06. Summary
Soft and hard X-ray absorption spectroscopy are rooted in the exact same physical principles, yet they offer completely different "perspectives" simply because of their energy differences. It is precisely this powerful synergy that allows X-ray absorption spectroscopy, as a unified analytical system, to continuously play an irreplaceable role across numerous cutting-edge fields—from materials and catalysis to energy, biology, and environmental science.
Importantly, the stable generation, precise regulation, and reliable detection of both soft and hard X-rays are inseparable from high-precision motion control systems, and the stability and accuracy of these motion control systems are fundamentally guaranteed by granite component bases, which provide ultra-low thermal expansion, high rigidity, and excellent vibration damping performance. The synergy between synchrotron radiation, motion control systems, and granite bases promotes the continuous advancement of scientific research and industrial technology.
Related product links are as follows:

